 
The total number of spherical nodal surfaces within s orbital of any shell (n) is equal to (n-1). This 2s orbital differs from 1s orbital in having a nodal sphere within it. This spherical shell is called a ‘Node’ or ‘Nodal Surface’. In 2s orbital, there is a spherical shell where the probability of finding the electron is almost zero. Therefore, 2s orbital is larger in size than 1s orbital. Graeter the value of ‘n’, larger is the size of orbital. The size of s orbital depends upon Principal Quantum Number (n). The only three-dimensional figure having one orientation in space is a sphere, hence, s orbital is spherical in shape. Thus, s orbital has only one orientation. On the basis of Quantum Numbers- For s orbital, l = 0 and m = 0, (l = Azimuthal Quantum Number and m = Magnetic Quantum Number). 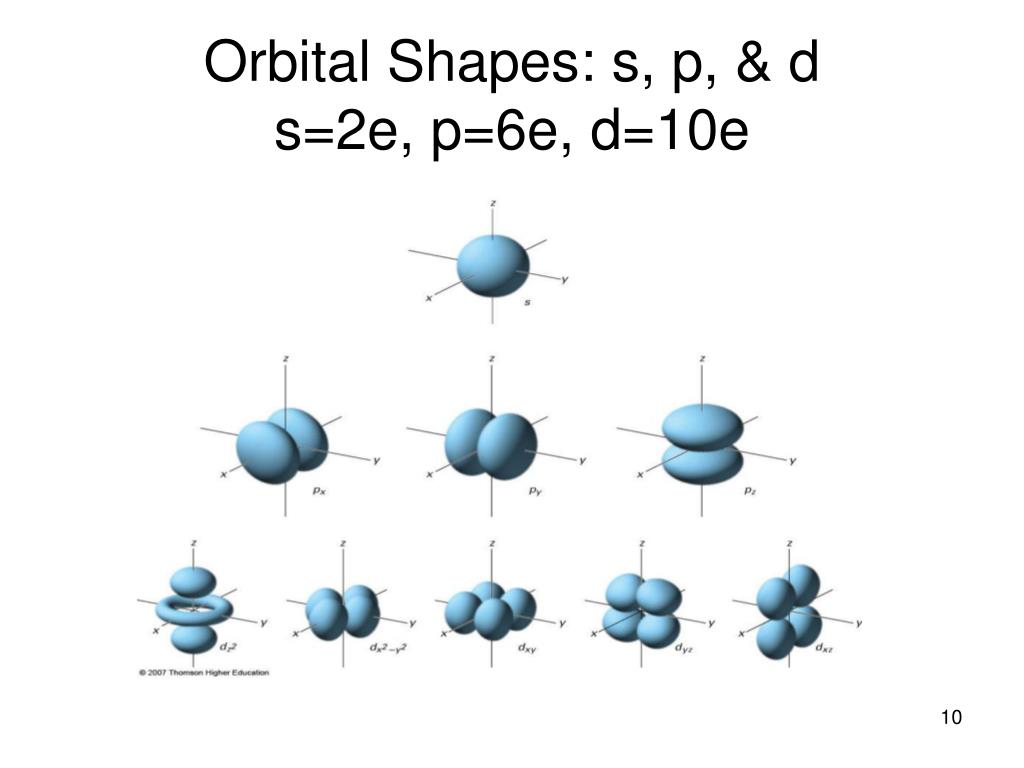
Hence, s orbitals are non-directional and spherically symmetrical about the nucleus, since a sphere can be defined completely by a single value.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
